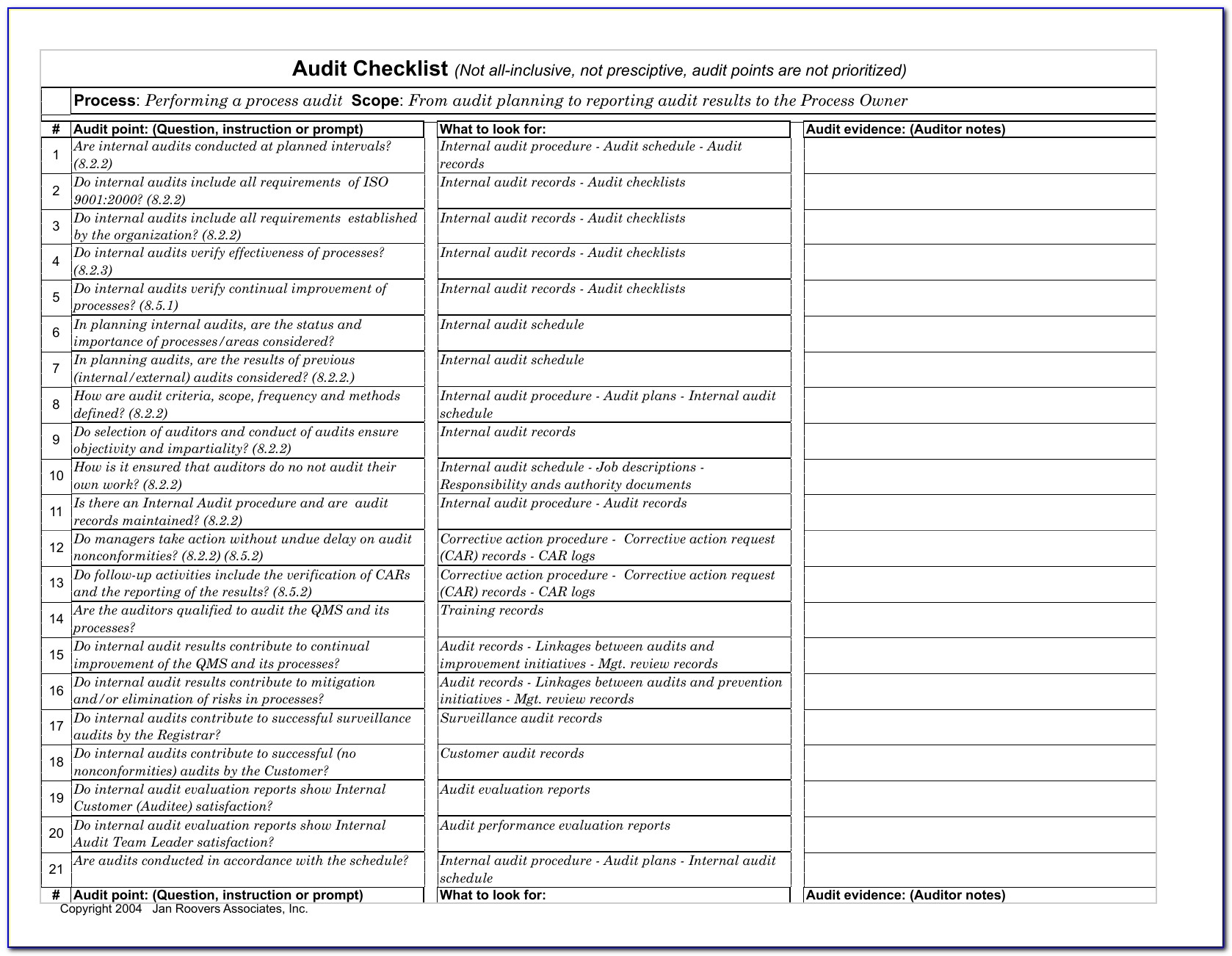
Evidence that the above points have been discussed – such as in minutes of management review meetings.The evidence you will need to look at during the audit will be: And that any new regulatory requirements that might impact the medical device are considered.That any recommendations for improving the medical device quality management system are communicated.Changes (legislation, organisation, technology etc) are reviewed.Follow up actions from previous reviews.Status of preventive and corrective actions are reviewed.Results of product testing/product release is reviewed – what monitoring is taking place.Results from processes are reviewed – are they continuing to work effectively.Results from internal audits will be reviewed.
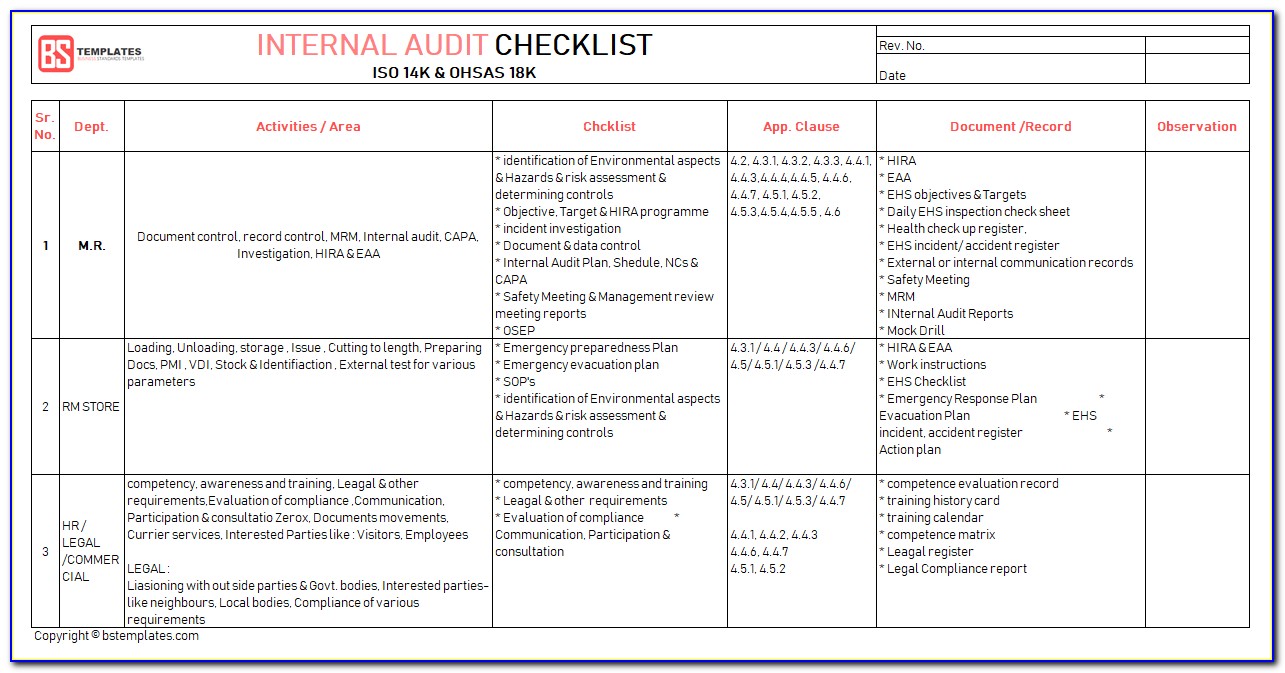
Any communications from the regulatory body (such as the MHRA in the UK) are considered.Results of records of feedback on the product are reviewed.If we take a look at clause 5.6, the key requirements are that: The overall intent of management review is to ensure the management system is implemented and effective in driving improvement. To give a simple example I have chosen Management Review from ISO 13485:2016 (Section 4.6) but this technique can be applied to any ISO standard including Harmonised Standards. Identify what objective evidence you are going to look for to prove the system works – would you expect to see records etc to demonstrate conformance.Breakdown the specific requirements of the clause of the standard (the ‘shalls’).The intent of the section/clause of the standard – knowing what the clause is aiming to achieve.The key steps to writing a checklist based on any management system standard is to understand:
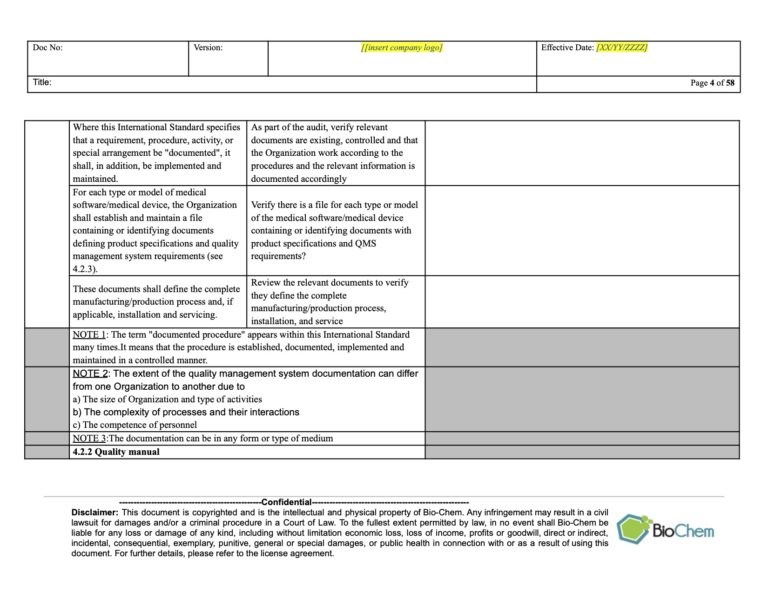
How to write an audit check list using an ISO standard This core skill provides you with a clear set of questions to ask during the audit, identifies what documents you might want to sample and keeps you on track with the audit timetable and objectives. One of the key audit skills we develop with delegates during our audit courses is the ability to write a good audit checklist.
